End of the One-Company Era in Soft Tissue Robotics
Hospitals are finally gaining choice as new entrants deliver versatile, high-performance soft tissue robotic platforms.
In an era defined by an overflow of real-time data, investment research often suffers from a paradox of choice. We have more technical indicators than ever, yet our fundamental insight has rarely felt more diluted. There is a tendency to over-index on granular ratios and immediate price action, often at the expense of recognizing paradigmatic shifts, the moments when an industry fundamentally breaks from its past.
We are witnessing massive structural realignment in the soft tissue robotic surgery market in real time. For over two decades, one pioneer defined this sector. Now the one-company era is rapidly dissolving into a multi-polar competitive landscape.
To properly understand the magnitude of this shift, I want to revisit where it all started. The concept of robotic surgery has its roots in military research aimed at solving a critical problem, allowing surgeons to operate on wounded soldiers remotely on the battlefield. The U.S. Defense Advanced Projects Agency (DARPA) funded research at SRI International in the late 1980s to create a telepresence system, which became the prototype for modern surgical robotics. While early commercial systems like the PUMA 560 (1985) and the voice-controlled AESOP (1994) introduced robotics to the operating room, the true inflection point occurred when the military’s core technology was commercialized. Recognizing the immense potential of the SRI system, Dr. Federic Moll partnered with John Freund and Robert Younge to acquire intellectual property, leading to the founding of Intuitive Surgical in 1995.
The founders rapidly developed their signature product, the Da Vinci Surgical System, which received U.S. FDA approval in 2000 for general laparoscopic procedures. This marked the birth of the modern surgical robotics market. Intuitive cemented its monopoly in 2003 by merging with its primary rival, Computer Motion, Inc. (makers of the ZEUS robotic system), effectively eliminating direct competition. Having established Da Vinci as the standard of care in soft tissue specialties, Intuitive Surgical has maintained undisputed global market leadership for more than two decades.
Intuitive’s journey to dominance paved the way for robotic surgery to become an integral part of modern surgical practice. If you look back to 1995, when the company was founded, the idea that a surgeon could operate from a console and feel the push and pull of tissue with such high fidelity was a distant fantasy. Today, robotic surgery has become a standard practice, adopted by leading hospitals and medical schools globally. This monumental shift is visible on the company’s vast scale. Intuitive now has an installed base of over 10,600 robotic systems across the globe, with over 17 million procedures performed to date.
What I find so remarkable is how far they have pushed the limits from that original military vision. Intuitive’s latest system, the Da Vinci 5, represents a staggering leap forward, delivering 10,000 times the computing power of its predecessor. It introduces revolutionary Force Feedback technology (also called haptic feedback) that finally allows the surgeon to feel subtle resistance, push, and pull forces on tissue through the console controls. This ability to deliver “enhanced surgical senses” is not just about feeling; it translates into tangible patient benefits, resulting in up to 43% less force exerted on tissue in preclinical trials. Furthermore, this computational muscle powers advanced digital features, giving surgeons real-time surgical sets and data analytics, such as the Force Gauge (a speedometer-like indicator of applied force), truly setting the standard for the future of surgery.
Hospitals are performing more robotic surgeries than ever, UCSF Health alone passed 15,000 robotic procedures in 2024.Via Christi hospitals in Kansas reported having performed more than 25,000 robotic‑assisted surgeries across their network as of October 2025, but even with all that momentum, the financial weight of robotic programs has not gone away.
These systems still come with multi-million-dollar price tags, and hospitals today are being forced to think more carefully about every dollar they spend, especially as the reimbursement landscape continues to tighten. So, you end up with this strange moment where the demand for robotics is surging, the technology is accelerating, and yet the economic pressures are becoming harder to ignore. This is where I personally see a huge shift coming, where competitors are going to experience a level of demand they have never seen before. Hospitals need more affordable options, promising new systems are finally making real progress in the U.S. and abroad, and it feels like we are just at the very beginning of what could be a major reshaping of the market.

The company I want to address first is SS Innovations (NASDAQ: SSII) as it brings the story into a full circle. Remember when I mentioned Dr. Frederic Moll, the co-founder of Intuitive Surgical, and the person widely regarded as the father of surgical robotics? He now serves as Vice-Chairman of SS Innovations and holds approximately 10% of the company.
What draws me the most is what they are trying to accomplish. The company is led by Chairman and CEO Dr. Sudhir Srivastava, a pioneering robotic cardiac surgeon who has performed and trained teams in many of the world’s first robotic heart procedures. Srivastava’s approach is not about replicating Intuitive Surgical’s platform, but about fundamentally rethinking how robotic systems are designed and deployed to improve access and usability. As quoted by MedTech Dive, Srivastava said, “My whole goal was always not to copy. Although [Intuitive] created a wonderful technology, there are always opportunities to make things better. Newer ways are being discovered, and we can use our thought processes to advance it further.”
Since their NASDAQ debut on April 25th 2025, the momentum behind the company has only intensified. They have officially submitted their 510(k) application on December 5, 2025, putting them on track for potential U.S. clearance as early as mid-2026. As of December 31, 2025, their core robotic system, SSi Mantra already has 168 installed across 10 countries, with more than 7,800 procedures performed. The main distinction is their presence in areas where other systems have not yet become a major factor, specifically cardiac and telesurgery. They have already recorded 400 cardiac cases and 120 telesurgeries.
I want to pause right here because what I just mentioned is nothing short of revolutionary. If you aren’t familiar with telesurgery, it is the ability for a surgeon to operate on a patient miles, or even continents, away via a robotic interface and a high-speed data connection. This concept has been a dream since the 2001 “Lindbergh Operation,” but it is only now becoming a clinical reality. SS Innovations is not the only company with regulatory approval in this field. MicroPort MedBot has received NMPA approval in China and completed nearly 400 cases within its domestic network. However, SS Innovations has uniquely expanded the scope of the technology by successfully performing the world’s first remote cardiac surgeries, a specialty requiring extreme precision.
What truly sets them apart is the SSi Mantra Tele Surgeon Console (TSC), the world’s first portable tele-surgeon console, that allows surgeons to operate entirely outside of a traditional hospital and OR setting. By utilizing lightweight 3D glasses and magnetic controls, specialists can now provide life-saving care from a private office or even their own residence, as demonstrated by their CEO’s recent 185-mile bypass surgery. SS Innovations made history when they officially received regulatory approval for the telesurgery and teleproctoring capabilities of the SSi Mantra from India’s CDSCO back on December 10, 2024. While the industry has been evolving for years, if they secure FDA approval for telesurgery applications, it will be monumental, opening the floodgates for remote, expert-led care to scale globally.
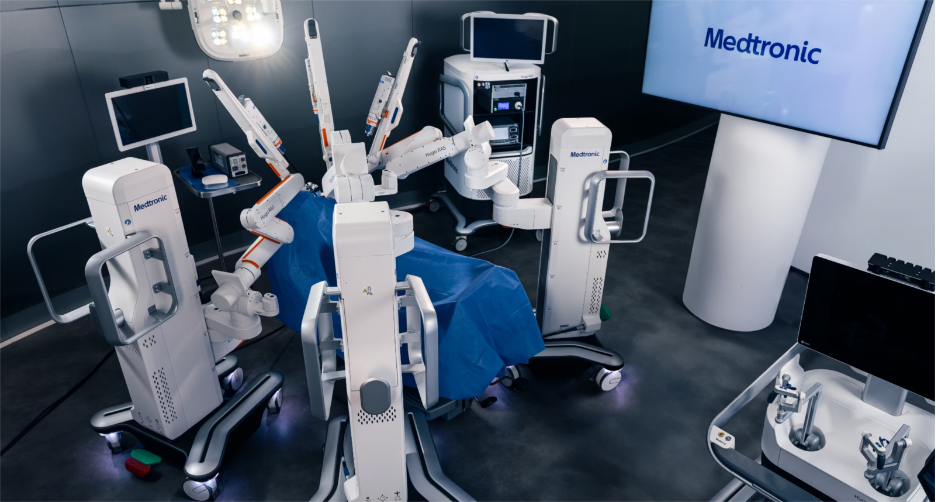
Medtronic, (NYSE: MDT) a MedTech conglomerate based out of Dublin, Ireland has recently made a huge regulatory stride. whose timing in the market says a lot about where this field is headed. When a company generating over $33 billion in annual revenue leans further into robotics, there is a level of deep-rooted integrity and scale that smaller competitors simply cannot replicate. Medtronic began rolling out its Hugo™ robotic system in 2021, steadily building an international presence while preparing for its eventual move into the U.S. market. In just a few years, Hugo secured clearances in more than 30 countries, creating a broad clinical foundation long before crossing into the United States.
On December 3, 2025, the FDA cleared Medtronic’s Hugo (RAS) for urologic procedures. Hospitals now have access to a robotic platform backed by one of the world’s most established medical technology companies. Hugo’s modular architecture and deep integration with Medtronic’s Touch Surgery™ digital ecosystem reinforce how serious of a competitor they are.
Personally, I do not believe investors are fully pricing in how significant this U.S. clearance is. For a company of Medtronic’s size and reputation, reaching this milestone marks a major achievement, and it sets the stage for an adoption curve that is going to be fascinating to watch over the next decade.

Johnson & Johnson (NYSE: JNJ) is also preparing an entrance into the market with the OTTAVA™ Robotic Surgical System, though it has not yet been commercialized. The company recently submitted the system to the U.S. Food and Drug Administration (FDA) for De Novo classification, seeking approval for multiple upper abdominal general surgery procedures, including gastric bypass, gastric sleeve, small bowel resection, and hiatal hernia repair. They have also reached a key milestone by completing its first clinical trial cases with the OTTAVA™ system on April 14, 2025, marking its initial use in real-world surgical procedures.
OTTAVA is designed as a multi-specialty soft-tissue platform capable of handling complex procedures across the upper abdomen. Its unified architecture and integration with Ethicon surgical instruments provide surgeons with familiar tools for minimally invasive procedures, potentially helping to accelerate adoption.
Just like Medtronic, this is another juggernaut of a corporation tapping into the soft-tissue robotics market. Although the companies I’ve highlighted so far are already commercialized and have multiple systems deployed in hospitals, Johnson & Johnson’s scale, resources, and expertise, in my view, solidifies that they will make significant strides once they receive authorization.

If you have noticed, the first three companies I have highlighted all have something in common besides being in the same industry, they are all publicly traded. In contrast, CMR Surgical is a fierce competitor in the private markets. Headquartered in Cambridge, United Kingdom, they have already achieved the milestone of becoming the second most adopted soft-tissue robotic system globally, positioned only behind Intuitive Surgical. Across all CMR’s robotic platforms, they have completed over 40,000 procedures, building a robust foundation.
They have made significant regulatory strides in the U.S. recently; after clearing the initial hurdle with a De Novo authorization in late 2024, they secured a critical FDA 510(k) clearance for the Versius Plus on December 16, 2025.
Back on April 2, 2025, CMR closed a $200 million funding round supported by major backers including a substantial debt investment from Trinity Capital, which brought their total capital raised to approximately $1.4 billion. Given the recent momentum in the IPO markets, the next step could very well be a public offering.

Finally, another competitor in the private markets is Moon Surgical, headquartered in both Paris and San Francisco. Dr. Frederic Moll serves as the Chair of the Moon Surgical board in addition to his role at SS Innovations. His presence on the board serves as a massive endorsement of Moon’s vision to bring robotics to the 18.8 million annual procedures currently ignored by larger telerobotic systems.
The company has moved at a breakneck pace, receiving FDA clearance for the commercial version of its Maestro™ Robotic Surgery System in June 2024. Most recently, in March 2025, they reached a major industry milestone with the FDA clearance of ScoPilot™, the first AI-enhanced intraoperative capability to run natively on a surgical robotics platform.
Powered by NVIDIA Holoscan, this physical AI allows the laparoscope to automatically follow a surgeon’s instrument tip, a breakthrough that has already supported the treatment of over 1,100 patients across the U.S. and Europe. By utilizing real-time sensing to enhance surgeon autonomy and OR efficiency, Moon Surgical is positioning itself at the absolute forefront of the digital surgery revolution.
What to expect going forward is a complete departure from the status quo. For twenty years, the soft tissue robotic industry was a one-act play. Now, we are seeing a diverse cast of competitors, from established medical giants to specialized global firms, reimagining what these systems can do. The technology has reached a point where it is a standard of care for many specialties, however its full potential remains widely untapped. Growth Global Insights predicts the global surgical robotics market will surge to more than $66.46 billion by 2035.
Looking ahead to the next 5, 10, and 15 years, the investment potential here is as significant as any other corner of technology. This field is evolving into an essential global infrastructure. While I do not expect a radical crash in costs, the entry of more versatile players will naturally introduce more flexible financial models and a gradual downward pressure on the total cost of ownership. As these newer platforms refine their capabilities and lower the barrier to entry, they are not just fighting for existing market share, but the entire pie. We are going to see businesses that are currently building their foundations to scale into heavyweights in a fast-growing industry.


Fascinating read and super insightful. Great work, Luke.
Excellent research and well written! You are a very talented writer!